Last verified: April 2026
Discovery Timeline: From THC to the Endocannabinoidome
The endocannabinoid system was discovered backwards — the exogenous ligand was identified three decades before the endogenous one. In 1964, Raphael Mechoulam and Yechiel Gaoni at the Weizmann Institute in Israel isolated and synthesized Δ9-tetrahydrocannabinol (THC) from Cannabis sativa, establishing for the first time the specific molecule responsible for cannabis psychoactivity. But THC’s mechanism of action remained unknown for another 24 years.
The breakthrough came in 1988, when Allyn Howlett and William Devane at St. Louis University demonstrated specific, saturable, stereoselective binding sites for cannabinoids in rat brain membranes using the synthetic cannabinoid CP55,940 as a radioligand. This was the first direct evidence that cannabinoids acted through a receptor — not through nonspecific membrane disruption, as had been widely assumed. In 1990, Lisa Matsuda and colleagues at the National Institute of Mental Health (NIMH) cloned the receptor, designating it CB1 — the first cannabinoid receptor characterized at the molecular level. Three years later, in 1993, Sean Munro’s group at Cambridge’s Medical Research Council (MRC) cloned a second receptor, CB2, from a human promyelocytic leukemia cell line (HL-60), establishing that the cannabinoid system extended beyond the brain into the immune system.
If receptors exist, the body must produce its own ligands. In 1992, Mechoulam, Devane, and Lumír Hanuš isolated anandamide (arachidonoyl ethanolamide) from porcine brain tissue. They named it after the Sanskrit word ananda, meaning “bliss” — a lyrical choice that has been both celebrated and criticized. In 1995, Shimon Ben-Shabat, working in Mechoulam’s lab, identified 2-arachidonoylglycerol (2-AG), which turned out to be far more abundant and would prove to be the primary endocannabinoid at both CB1 and CB2.
The field has expanded considerably since. Vincenzo Di Marzo has proposed the concept of the “endocannabinoidome” — a broader network encompassing not just the classical receptors and ligands but also related lipid mediators, orphan receptors (GPR55, GPR18, GPR119), ion channels (TRPV1, TRPA1), and metabolic enzymes that collectively form an extended regulatory network far more complex than the original CB1/CB2/anandamide/2-AG framework suggested.
Retrograde Signaling: Running the Circuit Backwards
The most distinctive feature of endocannabinoid signaling is that it operates in reverse. In classical neurotransmission, signaling molecules travel from the presynaptic neuron to the postsynaptic neuron. Endocannabinoids do the opposite: they are synthesized on demand in the postsynaptic neuron in response to depolarization or receptor activation, then travel backward across the synapse to bind CB1 receptors on the presynaptic terminal.
This retrograde mechanism gives the postsynaptic neuron a way to regulate its own inputs. When a postsynaptic neuron is receiving too much excitatory or inhibitory input, it synthesizes endocannabinoids that cross the synapse and activate presynaptic CB1, which suppresses further neurotransmitter release. The two best-characterized forms of this short-term plasticity are depolarization-induced suppression of inhibition (DSI) and depolarization-induced suppression of excitation (DSE), first described in hippocampal and cerebellar circuits.
This “circuit breaker” function is fundamental. The ECS does not initiate signals — it modulates them. It acts as a homeostatic brake, dampening excessive neuronal firing in both excitatory (glutamatergic) and inhibitory (GABAergic) circuits. This is why cannabinoid pharmacology is so context-dependent: the same compound can have opposite effects depending on which circuit it modulates.
Homeostasis: The Master Regulator Hypothesis
The endocannabinoid system has been described as a “master regulator of homeostasis” — a characterization supported by its involvement in an extraordinary range of physiological processes. ECS signaling has been documented in:
- Appetite and energy metabolism — CB1 activation in the hypothalamus and gut stimulates feeding behavior; rimonabant (a CB1 inverse agonist) caused weight loss but was withdrawn due to psychiatric side effects
- Pain modulation — endocannabinoids modulate nociceptive signaling at spinal, supraspinal, and peripheral levels
- Immune regulation — CB2 on immune cells modulates cytokine release and inflammatory responses
- Stress and anxiety — amygdalar and prefrontal endocannabinoid signaling regulates the hypothalamic-pituitary-adrenal (HPA) axis
- Neuroplasticity and memory — hippocampal CB1 modulates long-term potentiation and extinction of aversive memories
- Sleep — anandamide levels fluctuate with circadian rhythm and increase during sleep deprivation
- Bone metabolism — CB2 on osteoblasts and osteoclasts regulates bone remodeling
- Reproduction — endocannabinoids influence implantation, placentation, and spermatogenesis
The breadth of ECS involvement is not surprising when you consider its evolutionary age. John McPartland has traced endocannabinoid-like signaling components across 600 million years of evolution, identifying homologous elements in organisms as distant as sea squirts (Ciona intestinalis). The system predates the vertebrate nervous system itself.
Why Cannabis Cannot Cause Fatal Overdose
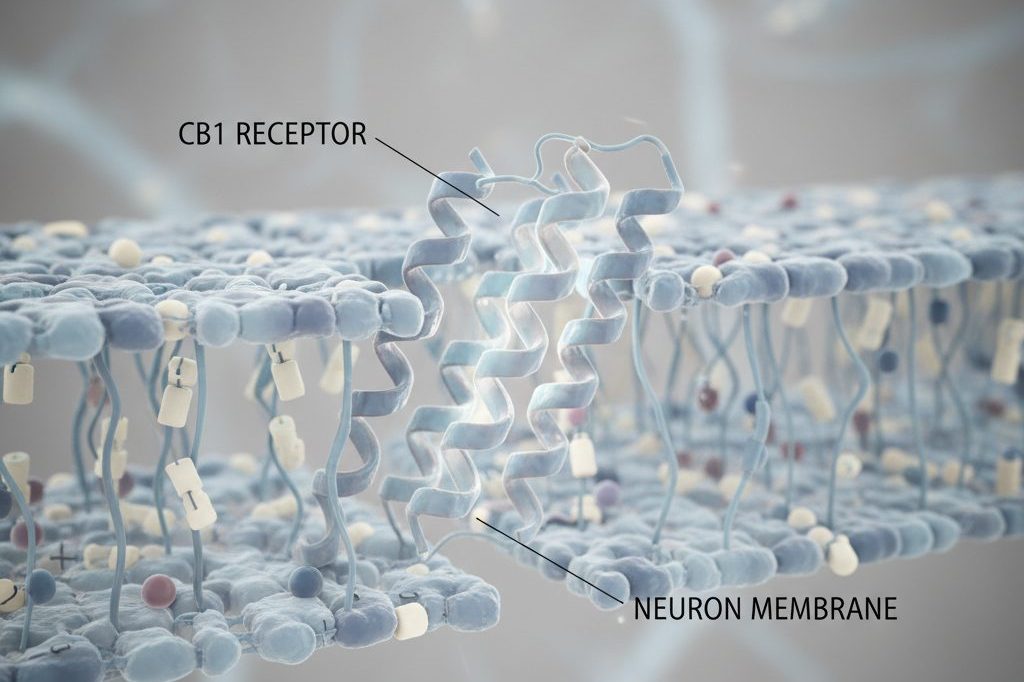
The distribution of CB1 receptors explains one of cannabis’s most notable safety features. CB1 is the most abundant G protein-coupled receptor (GPCR) in the human brain, with particularly high density in the basal ganglia (motor control), cerebellum (coordination), hippocampus (memory), amygdala (emotional processing), and cerebral cortex (higher cognition). These distributions explain cannabis’s characteristic effects: motor impairment, altered coordination, short-term memory disruption, emotional modulation, and perceptual changes.
Critically, however, CB1 receptor density is extremely low in the brainstem — the region controlling cardiovascular and respiratory function. Miles Herkenham’s autoradiographic mapping studies at NIMH in the early 1990s demonstrated this paucity clearly. Because brainstem cardiorespiratory centers lack significant CB1 expression, even massive THC doses cannot produce the respiratory depression that makes opioid overdose lethal. This is the anatomical basis for cannabis’s therapeutic index of approximately 1:20,000 to 1:40,000 — compared to roughly 1:6 for opioids and 1:10 for alcohol.
This does not mean cannabis is without risk. Cardiovascular effects (tachycardia, orthostatic hypotension), psychiatric effects (acute psychosis, anxiety), and the risk of cannabis use disorder are all well-documented. But fatal pharmacological overdose from cannabis alone is essentially impossible in humans, and this is directly attributable to CB1 receptor distribution.
Beyond CB1 and CB2: The Expanded Receptor Landscape
The classical two-receptor model is increasingly recognized as incomplete. Several additional targets are now considered part of the extended endocannabinoid system:
- GPR55 — sometimes called the “third cannabinoid receptor,” activated by lysophosphatidylinositol (LPI) and some cannabinoids; expressed in brain, gut, adrenal glands, and immune tissue; implicated in bone metabolism and cancer cell proliferation
- GPR18 — activated by N-arachidonoyl glycine (NAGly) and resolvin D2; expressed in immune cells; implicated in microglial migration and inflammation resolution
- TRPV1 (vanilloid receptor 1) — the capsaicin receptor; activated by anandamide at higher concentrations; involved in pain, thermoregulation, and neurogenic inflammation; represents a convergence point between endocannabinoid and vanilloid signaling
- PPARγ (peroxisome proliferator-activated receptor gamma) — a nuclear receptor activated by several cannabinoids; regulates lipid metabolism, insulin sensitivity, and anti-inflammatory gene transcription
This expanded receptor landscape is why individual cannabinoids produce such complex, sometimes paradoxical effects. THC, CBD, anandamide, and 2-AG each interact with different subsets of these targets at different affinities, and the net physiological outcome depends on the receptor expression profile of the target tissue, the local concentration of the ligand, and the presence of competing endogenous molecules.
The endocannabinoid system is perhaps the most important physiologic system involved in establishing and maintaining human health.
Dustin Sulak, DO — a widely cited characterization, though some researchers consider it hyperbolic given the equally critical roles of the autonomic nervous system and HPA axis
For in-depth cannabis education, dosing guides, safety information, and research summaries, visit our partner site TryCannabis.org
Related on this site: Clinical Endocannabinoid Deficiency, CB1 Receptor, Send Us a Message.
